Objectives:
1. To synthesize azo dyes
2. To understand the formation of azo dyes
3. To understand how to prepare a dye
Materials:
Sodium nitrite, concentrated HCl, sodium hydroxide solution, sodium chloride, 4-nitroaniline, salicylic acid, white cotton fabric
Apparatus:
Test tube, ice bath, vacuum filtration apparatus, glass funnel, and hotplate
Procedures:
Results and calculations:
Table 1: Observation of dye colour changes
| Observations | |
| During synthesis | When the darkish green slurry is added into hydrochloric acid, the mixture turns to darkish red. |
| During dyeing | The cotton fabric is dyed with darkish red but fades to pale brown. |
Table 2: Mass of solid dye
| Mass of filter paper | 0.3278g |
| Mass of (filter paper + solid dye) | 1.9518g |
| Mass of solid dye | 1.6240g |
Calculating percentage yield
Mole number of 4-nitroaniline = 0.7066g/ 138.12g mol-1
= 0.0051 mol
Mole number of sodium nitrite = 0.3800g/ 69g mol-1
= 0.0055mol
Thus, 4-nitroaniline is the limiting agent in the first reaction.
Mole number of salicylic acid = 0.68g/ 138.12g mol-1
= 0.0049 mol
In the second reaction, salicylic acid is the limiting agent.
Theoretical mole number of diazo compound = 0.0049mol
Actual mole number of diazo compound = 1.6240g/ 287.12g mol-1
= 0.0057mol
Percentage yield = 0.0057 mol/ 0.0049mol x 100%
= 116.33%
Discussion:
The purpose in this experiment is to synthesize azo dye and dye it on a cotton fabric. The colour of azo dye formed in this experiment was darkish red. However, it faded to pale brown after a few minutes. The amount of diazo compound obtained is 1.6240g with the percentage yield of 116.33%. The percentage yield is over 100% might be due to the presence of unreacted reagents.
In the synthesis of diazonium salt, sodium nitrite and 4-nitroaniline were mixed in water and then the slurry was added into concentrated hydrochloric acid. The darkish green solution was formed via the reaction as shown in diagram 1 below:
Diagram 1 Formation of diazonium salt
The azo dye was formed by further react with certain aromatic compound such as salicylic acid in this experiment via the process called coupling. The darkish red azo compound was formed as the following reaction of salicylic acid and diazonium salt.
The synthesized diazonium salt and azo compound showed their own colour because each compound contains aromatic ring and the aromatic allows the delocalisation of electron to occur. This delocalized electron system is capable to absorb different wavelength of light and hence each compound showed different colours.
Precaution steps:
1. Handle 4-nitroaniline carefully because it is highly toxic compound.
2. Avoid skin contact with sodium nitrite. It is toxic oxidiser.
3. Wear gloves when handling the dyes.


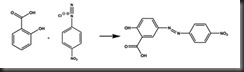
The synthesis of an azo dye requires two organic compounds- a diazonium salt and a coupling component.
ReplyDeleteBuy 6-APB Online
This comment has been removed by the author.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteDid a similar synthesis in lab using p-nitroaniline and 2-naphthol. Also got yield above 100%
ReplyDeletePlease Check Our New Products... Let's Join With Us http://www.sabungayam.biz/
ReplyDeleteTo find the actual mole number of the diazo compound, where did 287.12 come from?
ReplyDeleteMau Banyak Bonus?, Yuk Gabung Disini >> sabung ayam bali
ReplyDeleteMau yang lebih ????? ayam tarung
ReplyDeleteThanks.I have really learnt a lot from here
ReplyDeleteTips dan Trick Bermain Judi Online
ReplyDeleteAgen Toto Macau
Museumtoto IDNLive
Museumbola Slot IDN
Museumbola Slot Pulsa
Museumbola Joker123
AKSES SEGERA SITUS KAMI 1 ID BANYAK PERMAINAN
WA OFFICIAL : +6283157394921
ReplyDeleteYuk di add pin WA: +628122222995 taji ayam sabung
Sabung ayam online dan semua jenis permainan judi online ..
Semua bonus menarik kami berikan setiap hari nya ... :)
www,bolavita, ltd
Thanks for informative post. I am pleased sure this post has helped me save many hours of browsing other similar posts just to find what I was looking for. Just I want to say: Thank you! international certificate of good health (form 77-043) “You really know your stuff... Keep up the good work!”
ReplyDelete