Objective:
1. To determine the structural information on cinnamic acid prepared by using KBr disc technique
2. To identify an unknown sample by comparing it with the known sample
Introduction:
Infrared spectrometer (also known as spectrophotometer) is the instrument that used to determine the absorption spectrum for a compound. Two types of spectrometers are commonly being used in the analysis or research laboratory which includes dispersive and Fourier Transform (FT) infrared spectrometer. Both of these types of spectrophotometer provide the spectra of compounds in the common range of 4000 to 400 cm-1. Infrared region (IR) is divided into three regions which includes near IR, mid IR and far IR. The unit of IR measurement is wavenumber, the number of waves per centimeter, (cm-1). Although two provide nearly identical spectra for a given compound. FT infrared spectrometers provide the infrared spectrum much more rapidly than the dispersive spectrometers.
Infrared spectroscopy is resulted from the molecule vibration in which it includes stretching and bending. Every type of bond has a different natural frequency of variation and two of the same type of bond in two different compounds are in two slightly environments, two different structures have exactly the same infrared absorption pattern, or infrared spectrum. Although some of the frequencies absorbed in the two cases might be the same, but no identical infrared spectrum will appear in the two different molecules. Hence, infrared spectrum can be used for molecules much as a fingerprint can be used for human beings.
The main function of the infrared spectrum is to determine the structural information about chemicals either organic or inorganic. The functional groups that exist in a compound can be determined based on the spectra obtained. For example, infrared spectroscopy is also can be used to identify the chemicals from spills, paints, polymers, coatings, drugs and contaminants. The absorption of each type of bond is regularly found only in certain small portions of the vibrational infrared region. A small range of absorption can be defined for each type of bond. However, the absorptions are normally due to some other type of bond if outside this range. For an organic compound, it has a very rich and detailed spectrum while for an inorganic compound is usually much simpler.
In order to prepare a Kbr (potassium bromide) pellet, mortar and pestle, die set, sample holder and hydraulic KBr press are very important in preparation.
Mortar and pestle
Die set
Hydraulic KBr press
sample holder
A KBr pellet is a dilute suspension of solid in a solid. It sis usually obtained by first grinding the sample in anhydrous KBr at a ratio of approximately 1 part sample to 10-100 parts KBr. The mixture is then being placed on a steel plate containing a paper card with a hole punched in it. The sample is placed into the centre so that it will lie in the infrared beam when placed on the spectrometer. A second steel plate is placed over the sample and the steel sandwich is placed in a hydraulic press and subjected to pressures of 15000 psi. Decompression usually will give a KBr pellet that is reasonably transparent both to visible light and infrared radiation. The only limitation of KBr is that it is hydroscopic. This is usually a good idea to obtain a spectrum run as a Nujol mull with sample. Since Nujol mull is a hydrocarbon and has no affinity for water when compared to KBr. Any absorption in Nujol between 3400- 3600 cm-1 can be attributed to the sample and not to the absorption of water by KBr.
Materials: KBr powder, cinnamic acid, acetone, unknown sample, mortar and pestle
Apparatus: FTIR machine, hydraulic KBr press
Procedure:
Sample preparation by using KBr disc
1. The pure KBr powder was grinded by using mortar and pestle.
2. KBr was placed into the die set in which the sample is sandwiched by the steel plate with smooth surface.
3. The die set was compressed tightly by using a hydraulic KBr press.
4. The spectrum of pure KBr was obtained.
5. The cinnamic acid and KBr powder were mixed homogeneously and the spectrum of this mixture was obtained.
6. An unknown sample’s spectrum was obtained through KBr disc.
7. The spectrum of cinnamic acid and unknown were compared.
Results:
The spectrums of cinnamic acid and unknown compound, please refers to the spectrums obtained.
Discussion:
Potassium bromide (KBr) powder is always being used in the infrared spectroscopy analysis in which KBr does not absorb infrared region from 4000 cm-1 to 400 cm-1. KBr is transparent to IR radiation in the range of above 400 cm-1 and has no absorption bands in the region traditionally used for IR spectroscopy. The absorption bands recorded on the KBr disc are come from the sample or impurities present in the KBr mixture. The disadvantage of using KBr pellet in IR spectroscopy is due to its hygroscopic property. The KBr pellet absorbs the water that appears in the atmosphere and hence causes the existence of a broad band in the IR spectrum at around 3400cm-1 to 3200cm-1. In order to prevent this problem, the KBr powder must always being kept in the oven before it is being used in the mixing of sample. Besides, KBr disc is prepared in the solid state because this can prevent the reaction between sample and atmospheric contaminants or solvent to occur easily.
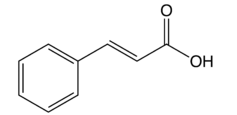
Every molecule will have its own characteristic spectrum. The bands that appear depend on the types of bonds and the structure of the molecule. The functional groups that present in the cinnamic acid involves –COOH and C=C bonds. Based on the spectrum obtained, the C=O double bond stretch exists at the wavenumber of 1681cm-1 due to the conjugation effect and hence it shifted to lower wavenumber. Besides, the –COOH functional group has a broad band O-H stretch and a C-O stretch with the wavenumber of 3100cm-1 ~ 2800cm-1 and wavenumber of 1200cm-1 ~ 1320cm-1 respectively. Since the cinnamic acid is polar molecule, the formation of dimer of carboxylic acid caused the O-H stretch shifted to from 3400cm-1~3300cm-1 to the current wavenumber of 3100cm-1~ 3000cm-1. Besides, the O-H stretch is actually overlapped the sp2 C-H stretch in which sp2 C-H should appears between 3100cm-1 ~ 3000cm-1. The two bands at 1418cm-1 and 1628cm-1 are represented by the aromatic C=C double bond in cinnamic acid. The aromatic C=C double bond is actually present between 1700cm-1 ~ 1500cm-1, but they shifted to lower wavenumber due to the conjugation effect of benzene ring. In addition, the band of aliphatic C=C double bond is overlapped by the aromatic C=C double bond stretch which actually present between 1680cm-1 ~ 1620cm-1 with a single peak (aromatic has two peaks). The figure 1 below shows the structural molecule of cinnamic acid:
Figure 1 Structure of cinnamic acid
Based on the spectrum of unknown compound, the very characteristic of the compound is the C=O double bond stretch near 1700cm-1. Since the C=O stretch is present at around 1700cm-1, no conjugation occurs in the C=O double bond. The band is very sharp because C=O is a strong absorber. The bond absorption takes place at 1604cm-1 indicated that the compound contains C=C double bond. This is an aliphatic compound instead of aromatic compound since there is absence of C=C stretch at 1400cm-1. Due to the presence of C=C double bond, the sp2 C-H stretch is also present in the spectrum which is indicated by the absorption band at 3056cm-1. This compound is not an amide although there is a peak present at 3417cm-1. This is because amide has a strong band in this IR region. The particular band with 3417cm-1 may be due to the contribution of other bonds. Based on the presence of C=O and C=C, the unknown compound is predicted as an aliphatic ketone with γ or higher degree of C=C double bond.
Precaution steps:
1. Make sure the mortar and pestle, die set and sample holder are being cleaned and washed with acetone to avoid contamination.
2. Make sure the KBr disc does not expose too long to atmosphere.
3. Make sure the mixture of KBr and sample are mixed homogeneously.
4. The die set must be placed in the centre of the hydraulic press in order to prevent it to be spoiled.



it is helpful topic. I really loved reading your blog. It was very well authored and easy to understand. Functional Near Infrared Spectroscopy
ReplyDeleteplz can u put the interpretation of DSC & IFTR spectrum
ReplyDelete