Characteristics
Pigment is a substance that changes its colour when exposed to visible light by reflecting or absorbing different wavelength of visible light. Pigments appear as the colours they are due to their wavelength-selective absorption properties. White light is the equal mixture of the entire spectrum of visible light with a wavelength range from about 400 nanometers to about 780 nanometers. When the pigment encountered by light, parts of the visible light are absorbed by the chemical bonds of the conjugated systems and other components of the pigment. At the same time, some of the visible light at other wavelength are reflected or scattered.
Other than that, most of the pigments are exist as polar compounds. They are not soluble in water like dyes but they are tend to soluble in the oil or other polar solvent.
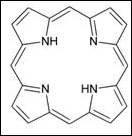
Pigments can be made up from a few to even more than ten benzene rings which attached with many different substituents in a single pigment molecule. Some of the pigments are made up from a porphyrin with a metallic element present in the centre. These pigments are called heme or porphyrin-based pigments. Diagram 1 shows the simplest structure of porphyrin which is one of the components in pigments. Examples of heme or porphyrin pigment are haemoglobin, myoglobin and hemocyanin.
Diagram 1 structure of porphyrin, the simplest structure of porphyrin
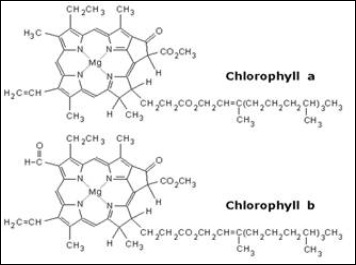
Furthermore, some of the biological pigments contain a long chain of hydrocarbon which attached to porphyrin-based. For example, chlorophyll is one of the examples that have a long chain hydrocarbon attached to a porphyrin-based. Diagram 2 shows the pigment structures of chlorophyll a and chlorophyll b.
Diagram 2 structure of chlorophyll
Organic pigments are considered as the substances that can imparts change of colour when expose to sunlight. This is because the presence of π electron conjugated system in organic compounds can absorb different wavelength of light to form many colourful dyes. Functional groups in organic pigment that absorb visible light is called chromophores such as -N≡N-, -C=C-, -C=O, -C=S, -C=NH, -N=O, -NO2. For inorganic pigments, the transition of electron from lower energy state to higher energy state induces the absorption of sunlight at certain wavelength and thus the complexes show colourful compounds. Hence, organic and inorganic pigments also possess a characteristic to exhibit different colours as their appearances.
Great article on characteristics of pigments and very well supported by chemical and molecular structures which makes more informative. Being an employee with Alliance Organics it feels pleasure to announce that Alliance Organics has become one of the renowned dyestuffs manufacturer and major players in terms of market share for exporting high quality organic pigments and inorganic pigments across a variety of countries like USA, China, Indonesia, Russia and Brazil.
ReplyDelete