Objectives
1. To understand the chemistry of activation energy.
2. To determine the activation energy for the reaction of bromide and bromate ions in acid solution.
Introduction
Activation energy is defined as the minimum energy barrier that must be overcome for a chemical reaction to take place. It is usually denoted as Ea, and given in unit of kiloJoule, kJ/mol. For a chemical reaction, an appreciable number of molecules with the energy equal to or greater than activation energy should be exist in the system. In order for a reaction to occur, the reactant particles must collide according to the collision theory. However, not all collision are able cause the reaction to happen, only a certain collisions in the system can cause chemical reaction, which is called effective collision. The effective collisions of molecules must collide with the correct orientation and sufficient energy to overcome the activation energy barrier. The energy is needed to break the existing bonds and form the new bonds of the molecules which resulting in the formation of products.
The activation energy of a reaction can be measured by using Arrhenius equation as shown in the equation below:
k = Ae-Ea/RT
where k = rate constant
T = absolute temperature
Ea = energy of activation
R = gas constant
The pre-exponential term, A is the property of particular reaction related to the collision frequency of the reactive species and thus is temperature dependent. However, according to the equation, the dependence of k on temperature is dominated by the strong exponential term, so the dependence of A on temperature is usually ignored as a first approximation. By taking logarithms of both sides,
Log10 k = -Ea / 2.303RT + log 10 A
= -Ea /2.303RT + constant
So, when a reaction has a rate constant that obeys Arrhenius equation, a plot of log10 k versus 1/T gives a straight line. The gradient of the straight line is –Ea / 2.303R while the interception of the straight line on the y-axis of the graph can be used to determine the values of log10 A.
Now, the rate of reaction is higher when the time taken for a fixed amount of reaction to complete is shorter. This makes the time taken, t to complete a fixed amount of reaction is inversely proportional to the rate constant, k.
T α 1/k
or t = constant/k
By taking logarithms of both sides,
Log10 t = - log10 k + constant
= Ea / 2.303RT + constant
A plot of log10 t versus 1/T gives a straight line as well and the slope of the graph is Ea / 2.303R. Thus, if t is measured at several temperatures then the energy of activation can be found.
In this experiment, the above method is applied to the reaction of bromide and bromate ions in an acid solution which occurs slowly at room temperature.
KBrO3 + 5 KBr + 3 H2SO4 --> 3 K2SO4 + 3 Br2 + 3 H2O
or BrO3- + 5 Br- + 6H+ --> 3 Br2 + 3 H2O
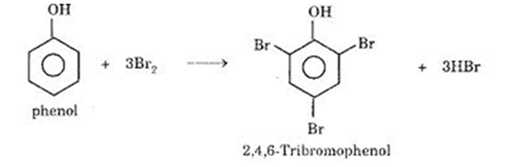
The time required for a fixed amount of the reaction to be completed, t is found by adding a fixed amount of phenol and some methyl red indicator to the reaction mixture. The bromine produced in the first reaction reacts very rapidly with the phenol to form tribromophenol.
C6H5OH + 3 Br2 --> C6H2Br3OH + 3 HBr
When all the phenol has reacted, the bromine continuously produced in the first reaction will then react with the methyl red indicator and bleaches its colour.
Methyl red + Br2 --> colourless compound
Apparatus : 1 dm-3 beaker, 3 100cm3 beakers, 2 boiling tubes, 1 5cm3 pipette, 1 10-cm3 pipette, thermometer (0 - 110°C), stopwatch
Material : 0.01 mole dm-3 aqueous phenol solution, bromide/bromate solution (0.0833 mole dm-3 potassium bromide and 0.0167 mole dm-3 potassium bromate, equivalent to 0.05 mole dm-3 bromine), 0.3 mole dm-3 sulphuric acid, methyl red indicator.
Procedures
1. 10 cm3 of phenol solution and 10 cm3 of bromide/bromate solution were pipette into one boiling tube.
2. Four drops of methyl red indicator were added to the mixture.
3. 5 cm3 of sulphuric acid was pipette into another boiling tube.
4. The two boiling tubes were immersed in the water bath of (75 ± 1) °C.
5. The contents of the two tubes were mixed by pouring rapidly from one tube to the other twice and the stopwatch was started at the same time.
6. The boiling tube containing the reaction mixture was kept immersed in the water.
7. The time required for the red colour of the methyl red indicator to disappear was determined.
8. The whole experiment was repeated at 65, 55, 45, 35, 25 and 15 °C.
9. Ice was used to achieve the lowest temperature.
Results & Calculations
(Assume R = 8.314 J K-1 mol-1)
From Graph 1,
Slope of the graph = Ea / 2.303R
Ea / 2.303R = (2.94 – 2.20) / (3.35×10-3 – 3.10×10-3)
Ea /2.303R = 0.74 / (2.50×10-3)
Ea = 2960 x 2.303R
Ea = 2960 x 2.303 x 8.314
Ea = 56676 J/ mol
Energy of activation, Ea = 56676 J /mol
Discussion
The reaction between bromide and bromate ions in acid solution is a slow chemical reaction at room temperature. This may be due to the high activation energy of the reaction, which required 56.676 kJ of energy in order for a reaction to take place. According to collision theory, high activation energy will cause the product more difficult to form since it is not sufficient energy for that molecule collide without enough energy. Any reaction cannot occur if the colliding molecules do not have the energy equal or higher than its activation energy. There are also other factors that can reduce the effectiveness of collisions of molecules such as present or absent of catalyst.
According to Table 1, we can observed that the higher the temperature of the reactant species, the shorter the time taken for the disappearance of red colour of methyl red indicator. This is shows that shorter time taken in the reaction once the reaction is faster and higher rate of reaction. Although the activation energy for the reaction to occur remains unchanged at all the temperature, but the rate of reaction increased as the temperature increased. This means that the rate of reaction is depends on the temperature of the reactant. This has also been proven in Arrhenius equation where rate constant, log10 k is proportionally to 1/T with the slope of the graph, -Ea/R and a constant of log10 A.
Log10 k = -Ea/2.303RT + log10 A
The higher temperature caused the value of the -Ea/2.303RT closer to the value of 0. The constant log10 A will then minus off the value of -Ea/2.303RT and results in a larger value. The larger value will caused the value of the rate constant, k to become larger as well. Larger value of k will then results in faster reaction. Hence, this is proven that the higher the temperature of reactants, the reaction will proceed faster with the higher rate of reaction.
Besides, the rate of reaction roughly doubles for every 10 °C increase in temperature. This is because increase in temperature increase the kinetic energy of the molecules, the molecules with higher kinetic energy can move faster. With the higher speed, the molecules will collide more frequently results the larger amount of successful collision. With the higher kinetic energy of the molecules, the molecules can overcome the activation energy barrier during the collision and hence the reaction can be take places. As a result, the rate of the reaction could be increased if more heat energy is provided to the reactants.
Fixed amount of phenol and methyl red indicator were added to the mixture contents for the different temperature. This is because phenol can provides an intermediate state before the bromine molecules produced in the reaction between bromate and bromide ions in acid solution which is able to bleach the methyl red immediately. In other words, phenol is used to observe the time taken for the bromine molecules to react completely with phenol before bleach the methyl red indicator. The purpose of adding methyl red into the solution is to provide a colour which can easily to be observed. When the sulphuric acid was poured to the bromate/bromide ions solution, the methyl red indicator turns to pink colour. The bleaching effect from the bromine molecules caused the methyl red indicator to turn colourless after all the phenol is used up. This shows a colour changes which the time taken should be stopped. In order to compare the time taken for the bleaching of methyl red colour, the amount of phenol used in the repeated experiment must be equal.
The reason of choosing phenol as the reactant is due to it can form an intermediate state because phenol can react with bromine molecules quickly to produce tribromophenol and hydrogen bromide. This is happens before the bromine molecules react with the methyl red.
The –OH group in the phenol is an activating group in the benzene ring which can donates electron into the benzene ring to stabilize it. So, the product can be easily to form in the reaction. –OH group is ortho-para activating group in the benzene ring. This means that the incoming substituent will go into the ortho position or the para position, but hardly go into the meta position. This is due to the high energy is required for the product with a substituent at meta position to form in the reaction. The bromine molecules undergo substitution reaction in this reaction by substituting three hydrogen atoms from the benzene ring with three bromine atoms to maintain the aromaticity of the ring in phenol.
The reaction between bromate and bromide ions in acid solution is a redox reaction.
BrO3- + 5 Br- + 6H+ --> Br2 + 3H2O
The potassium and sulphate ions act as spectator ions in this experiment and they are not participate in any of the redox reaction or changing of their state. The bromide ions undergo oxidation by donating one of its electrons to the bromine atom in the bromate ions. The bromine atom in the bromate ions then undergoes reduction by receiving electron from the bromide ions. The hydrogen ions and oxygen atom in bromate ions does not involve in increase or decrease in oxidation number but they were involved in changing the state from the ions in aqueous solution to the water molecule in liquid state.
nice post bro.I was afraid of chemistry but you post made me to read the full post.This is so descriptive.Keep up your good work.
ReplyDeleteIt helps alot for my reports.. thanks man
ReplyDeleteThanks bro
ReplyDeleteits helped me a lot for my lab exam
hi i was just wondering how did you get the 2.303 when you took log of both sides?
ReplyDeleteThe actual logarithm used is the natural logarithm and since the ln value of a number is 2.303 times the value of the natural logarithm of it they divide the other side by 2.303 to compensate for this
Deletefantastic post! helped me a lot with my report, thank you so much! :)
ReplyDeletefound it very helpfull for my lab report....thanks
ReplyDeleteamazing v good post
ReplyDeleteDoes anyone actually have the time taken for each temperature
ReplyDeleteNice bro , too nice☝
ReplyDeletethanks...i appreciate your effort on the explanation, it's helpful
ReplyDeleteThank u bro for this
ReplyDeleteAlfa Chemistry offers an extensive catalog of building blocks, reagents, catalysts, reference materials, and research chemicals in a wide range of applications. N-octylpyridinium bromide
ReplyDeletethank you soo much
ReplyDeleteWhy methyl red becomes colourless after we mix sulforic acid with the mixture what's the reason ?
ReplyDeleteThanks alot bro, it really helped me with my lab report
ReplyDelete