Objectives:
- To synthesize n-butyl ethyl ether from 1-butanol
- To understand mechanism involved in the reaction
Introduction:
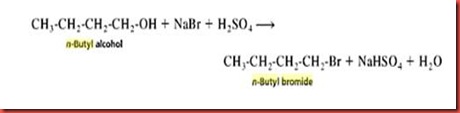
In this experiment, the procedure to generate n-butyl ethyl ether from 1-butanol is divided into two parts. The first part involves the formation of n-butyl bromide from 1-butanol. Alkyl halides are very useful intermediates in organic syntheses. The most common synthetic preparation of alky halides is the replacement of the hydroxyl group, OH of an alcohol by a halogen, HX. The displacement of a hydroxyl group by halide ion is successful only in the presence of a strong acid. 1-butanol is used to be converted into 1-bromobutane with adding of sodium bromide and sulphuric acid. The nucleophile for the reaction is Br- ions. The nucleophile in this lab is generated from an aqueous solution of sodium bromide. The sulfuric acid acts as a catalyst in this reaction. The sulphuric acid protonates 1-butanol to produce suitable leaving group, OH, in SN2 reaction. The chemical reaction is shown as below:
If this displacement reaction is attempted in the absence of an acid it is unsuccessful because leaving group would be a hydroxide ion which is a poor leaving group and a strong base.
In the second part of the experiment, the n-butyl bromide produced in the first part is being converted into n-butyl ethyl ether by using methanol and sodium hydroxide. The chemical reaction is shown as below:
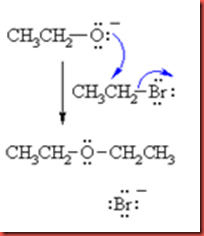
The mechanism of synthesis of ether is also known as mechanism of Williamson ether synthesis. This mechanism involves one alkoxide reacts with alkyl bromide to form ether with two alkyl groups by using a strong base. This reaction can be used to produce both symmetrical or unsymmetrical ethers and also cyclic ethers. The following diagram is the mechanism of the Williamson ether synthesis.
The alkoxide ion functions as nucleophile and attacks the electrophilic C of the alkyl halide, displacing the bromide and creating the new C-O bond.
For the synthesis of n-butyl ethyl ether, both reactions are undergoes SN2. SN2 is known as second order nucleophilic substitution which is for bimolecular process. The kinetic rate of SN2 is defined as
Rate = k [R-X][Nu-]
The more the alkyl groups attached to the reacting carbon, the slower the reaction. The order os reactivity in SN2 is shown in the following:
Tertiary alkyl > secondary alkyl > primary alkyl > methyl
--------------------------------->
Reactivity increasing
SN2 involves the inversion of configuration (rearrangement of atoms in molecule) to form a transition state which is different with SN1. First order nucleophilic substitution is a unimolecular process which form carbocation during the reaction.
Apparatus: Round bottomed flasks (50cm3 and 250cm3), Bunsen burner, condenser, thermometer, separating funnel
Materials: Sodium bromide, 1-butanol, conc. sulphuric acid, anti-bumping granules, 5% aqueous sodium bisulphate, distilled water, 10% aqueous sodium carbonate, anhydrous calcium chloride, sodium hydroxide, 95% ethanol, anhydrous magnesium sulphate
Procedure:
i) n-Butyl bromide
1. 27g of sodium bromide, 30cm3 of water and 20cm3 of 1-butanol are placed into a
250cm3 round bottom flask.
2. The mixture is cooled in an ice bath and 25cm3 conc. sulphuric acid is added with continuous swirling.
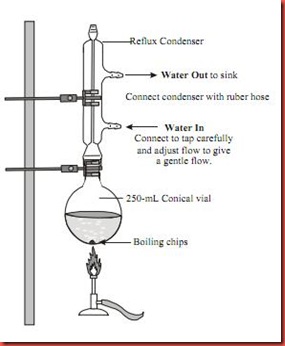
3. Two or three anti-bumping granules are added and attached with a gas trap to prevent HBr escaping, the round bottom flask is heated vigorously under reflux for 1.5 hours as shown in diagram 1 below.
Diagram 1
4. Distill the two layered mixture until the temperature reaches the boiling point of water.
5. The distillate is transferred to a separating funnel and shakes with an equal volume of 5% aqueous sodium bisulphate.
6. Allow the two layers to separate and wash the organic layer twice with 25cm3 water followed by 10% aqueous sodium carbonate (25cm3).
7. The product is dried with 5g calcium chloride and is filtered into 50cm3 round bottom flask.
8. Anti bumping granules are added and distilled, the material which boiled between 90-105 ̊C is collected.
9. The appearance of product is noted and weight is measured.
ii) Ethyl n-butyl ether
1. 4g of sodium hydroxide and 12cm3 95% of ethanol are added into round bottom flask and is heated under reflux for 20 minutes.
2. 10cm3 n-butyl bromide is added into the mixture through the top of the condenser and the reaction is heated under reflux for 90 minutes.
3. After cooling, the mixture is transferred to a separating funnel and 50cm3 of water is added which has been used to rinse the reaction flask.
4. The mixture is shaked and the lower layer is removed.
5. The washing is repeated for two times with 20cm3 of water.
6. The organic layer is dried with anhydrous magnesium sulphate and the liquid is filtered into a 50cm3 round bottom flask.
7. The product is distilled slowly; the material which boiled in the range 90-96 ̊C is collected in a pre-weighed flask.
8. The density of the pure ethyl n-butyl ether is determined by pipetting 1cm3 liquid into a pre-weighed measuring cylinder and noting the weight difference.
Results and calculation:
Part I
Weight of conical flask = 78.2260g
Weight of conical flask + weight of n-butyl bromide = 89.8416g
Weight of n-butyl bromide = 11.6156g
Density = Mass/Volume
Mass = (Volume x Density) / Molecular mass
Number of mole of 1-butanol = (20cm3 x 0.81g/cm3) / 74.08g mol-1
= 0.2187 mole
1 mole of NaBr reacts with 1 mole of 1-butanol to produce 1 mole of butyl bromide.
Thus, 0.2187 mole of butyl bromide is formed.
Theoretical weight of butyl bromide = 0.2187 mole X 136.972 g/mol
= 29.9558g
Experimental weight of butyl bromide = 11.6156g
Yield percentage of n-butyl bromide = 11.6156g/29.9558g X 100% = 38.78%
Part II
Weight of conical flask = 51.1825g
Weight of conical flask + weight of n-butyl ethyl ether = 58.0435g
Weight of n-butyl ethyl ether = 6.8610g
Weight of 5ml measuring cylinder = 14.7446g
Weight of 1ml n-butyl ethyl ether + weight of 5ml measuring cylinder = 15.6545g
Weight of 1ml n-butyl ethyl ether = 0.9099g
CH3CH2CH2CH2Br + NaOH + CH3CH2OH ---->
CH3CH2CH2CH2-O-CH2CH3 + NaBr + H2O
Number of mole of n-butyl bromide = (10 cm3 x 1.2686 g cm-3)/ 136.972g mol-1
= 0.0926 mole
Theoretical weight of n-butyl ethyl ether = 0.0926 mole x 102.112g/mol
= 9.4556g
Experiment weight of n-butyl ethyl ether = 6.8610g
Yield percentage of n-butyl ethyl ether = 6.8610g/9.4556g x 100% = 72.08%
Density = mass/volume
Density of n-butyl ethyl ether = mass of 1ml of n-butyl ether / volume of 1ml of n-butyl ether
= 0.9099g/1cm3
=0.9099g/cm3
Discussion:
Alkyl halides can be prepared from alcohols by reacting them with a hydrogen halide, HX (X = Cl, Br, I). The mechanisms of acid-catalyzed substitution of alcohols are termed SN1 and SN2, where “S” stands for substitution, the “N” stands for nucleophilic, and the “1” or “2” for unimolecular or bimolecular, respectively. The purpose of this experiment is to synthesize n-butyl ethyl ether via an SN2 reaction and to purify it using simple distillation where substances with different volatility and boiling points are separated from each other.
In the experiment, the primary alkyl halide n-butyl bromide can be prepared easily by allowing 1-butanol to react with sodium bromide and sulphuric acid. The sodium bromide reacts with sulphuric acid under reflux to produce hydrogen halides. The chemical reaction as shown in below indicates that the hydrogen halide is produced from the reaction.
2 NaBr + H2SO4 ------> 2 HBr + Na2SO4
The hydrogen halide produced is used to convert 1-butanol to become butyl bromide by undergoes nucleophilic substitution. Excess sulphuric acid serves to shift the equilibrium and thus to speed up the reaction by producing a higher concentration of hydrobromic acid. The sulphuric acid also protonates the hydroxyl group of 1-butanol so that water is displaced rather than the hydroxide ion OH-. The acid also protonates the water as it is produced in the reaction and deacticvates it(water) as a nucleophile, hence the water keeps the butyl bromide from being converted back into the alcohol by nucleophilic attack of water.
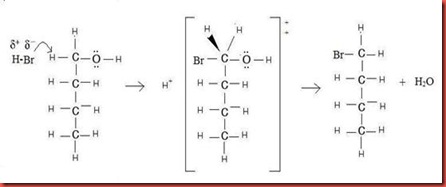
Synthesis of butyl bromide from the 1-butanol is undergoes SN2 mechanism. SN2 is known as second order nucleophilic substitution for bimolecule. The essential feature of the SN2 mechanism is that take place in a single step without intermediates when the incoming nucleophile, hydrogen bromide reacts with the 1-butanol from a direction opposite the group that leaves. As the bromide ion, Br- comes in on one side and bonds to the carbon, the OH- departs from the other side, thereby inverting the stereochemical configuration. The mechanism is shown in the figure 1 as below.
Figure 1
Since the hydrogen halide is polar molecule, the bromide ion is partial negative and the hydrogen is partial positive. The highly partial negative bromide ion acts as a nucleophile to attack the 1-butanol at the opposite side of the departing OH group. This leads to a transition state in which the new Br-C bond is partially forming at the same time the hold C-OH bond is partially breaking, in which the partial negative charge by both the incoming nucleophile and the leaving hydroxyl ions. The transition state for this inversion has the remaining three bonds to the carbon in a planar arrangement as shown in figure 1. The water is formed after the bromide successfully becomes part of the molecule.
After the reflux process, the distillate is introduced with same approximate same amount of sodium bisulphate, NaHSO4. The purpose of adding of sodium bisulphate is used to absorb water from, the organic solvent since it has high hydrophobic property. Sodium carbonate (mild acid) is added to neutralize the acidic solution. Anhydrous calcium chloride is added as drying agent to absorb water droplets in order to purify the organic layer. An excess drying agent should be used to ensure that all the water in solvent is removed. If the water remains in the materials collected, it could interfere with the analysis. After filter out the drying agent, several anti bumping granules (boiling chips) are added to prevent over boiling during distillation. Distillation process is carried out to purify the butyl bromide in the range of temperature 90°C to 105°C. The pure n-butyl bromide is obtained in the distillation process.
The n-butyl bromide formed after complete distillation in the first part is used as the materials in the latter part of this experiment. Now, the n-butyl bromide is used to synthesis n-butyl ethyl ether. The synonym of n-butyl ethyl ether is known as ethoxy butane which has the molecular formula with C6H14O. This molecule is categories in the ether group which has the general formula of R-O-R’. Sodium hydroxide and ethanol are introduced together and is heated under reflux. The purpose of introduction sodium hydroxide and ethanol is to produce ethoxide ions. The chemical reaction is shown as the following equation:
Figure 2
During reflux, the ethoxide ions react with n-butyl bromide under the second time reflux to form n-butyl ethyl ether as product. In this reaction, the bromide ions are escaped from the alkyl bromide and combine with the sodium ions to form sodium bromide to achieve stable molecule.
Figure 3
The butyl bromide undergoes second order nucleophilic substitution, SN2. The ethoxide functions as nucleophile which attacks the electrophilic C of the butyl bromide by displacing the bromide and creating a new C-O bond between ethoxide and butyl bromide. The bromide ion is being displaced and leaves the butyl group. As a result, an ether with butyl ethyl groups is formed which is known as n-butyl ethyl ether. The mechanism is shown in the figure 4 as below.
Figure 4
After the reflux, the mixture is transferred into a separatory funnel and water is introduced to rinse the mixture. This is because water is used to dissolve some unreacted NaOH and hence the NaOH can be removed by removing the water. In order to remove the water droplets, anhydrous magnesium sulphate is added. Distillation of the organic layer is carried out to purify the product. Pure n butyl ethyl ether is obtained through the distillation at temperature 90°C-96°C.The overall chemical equation for the synthesis of n-butyl ethyl ether is
CH3CH2CH2CH2Br + NaOH + CH3CH2OH ----->
CH3CH2CH2CH2-O-CH2CH3 + NaBr + H2O
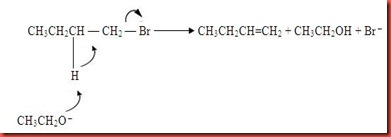
In the experiment, the but-1-ene is being produced via E2 elimination mechanism. E2 elimination reaction also can be occurred because the ethoxide ions are strong base which initiate elimination reaction to compete with the substitution reaction. The mechanism of elimination of butyl bromide is shown in figure 5.
Figure 5
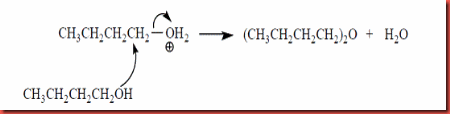
In figure 5, the mechanism shows that the bromide atom in butyl bromide attracts the electron from the C-Br bond to form partial negative and this causes the C1 lack of electron. The hydroxide ion acts as nucleophile and attack the electrophile C1 via E2 mechanism to form a transition state. As the OH- start to attack a neighboring H and begins to remove the H at the same time as the C-C double bond starts to form and the Br group start to leave. The water and bromide ion are leave and hence but-1-ene are formed via elimination. However, dibutyl ether also can be formed due to the strong sulphuric acid used. The strong acid causes the side reaction to the butyl alcohol which is dehydration and ether formation which is shown in the figure 6 below:
Figure 6
![clip_image002[5] clip_image002[5]](http://lh3.ggpht.com/_VE77yZI3fI0/TdZVW2OlaTI/AAAAAAAAAao/DUwnDxmnUbc/clip_image002%5B5%5D_thumb%5B2%5D.jpg?imgmax=800)
![clip_image031[5] clip_image031[5]](http://lh5.ggpht.com/_VE77yZI3fI0/TdZVe0IXkcI/AAAAAAAAAbY/pbAeCARgUQk/clip_image031%5B5%5D_thumb%5B1%5D.jpg?imgmax=800)
![clip_image033[5] clip_image033[5]](http://lh3.ggpht.com/_VE77yZI3fI0/TdZVge9b2ZI/AAAAAAAAAbg/zxwDVRGqXUU/clip_image033%5B5%5D_thumb%5B1%5D.jpg?imgmax=800)
![clip_image035[5] clip_image035[5]](http://lh5.ggpht.com/_VE77yZI3fI0/TdZVhu2kqlI/AAAAAAAAAbo/jg5yxTgz1Z0/clip_image035%5B5%5D_thumb%5B1%5D.jpg?imgmax=800)
You may search for our products through the search bar on our website. If you would like to receive a copy of our product catalog, please contact us at info@alfa-chemistry.com. N-hexylpyridinium bromide
ReplyDelete